The death toll of an outbreak linked to contaminated recalled eye drops has risen and more people have lost their vision.
According to an update issued by the Centers for Disease Control and Prevention on Tuesday, the number of deaths has risen from one — which occurred in Washington state — to three.
What’s more, at least eight people have gone blind and four people have had their eyeballs surgically removed.
The CDC did not provide any information in its update about the affected patients including names, ages, sexes or where they live.
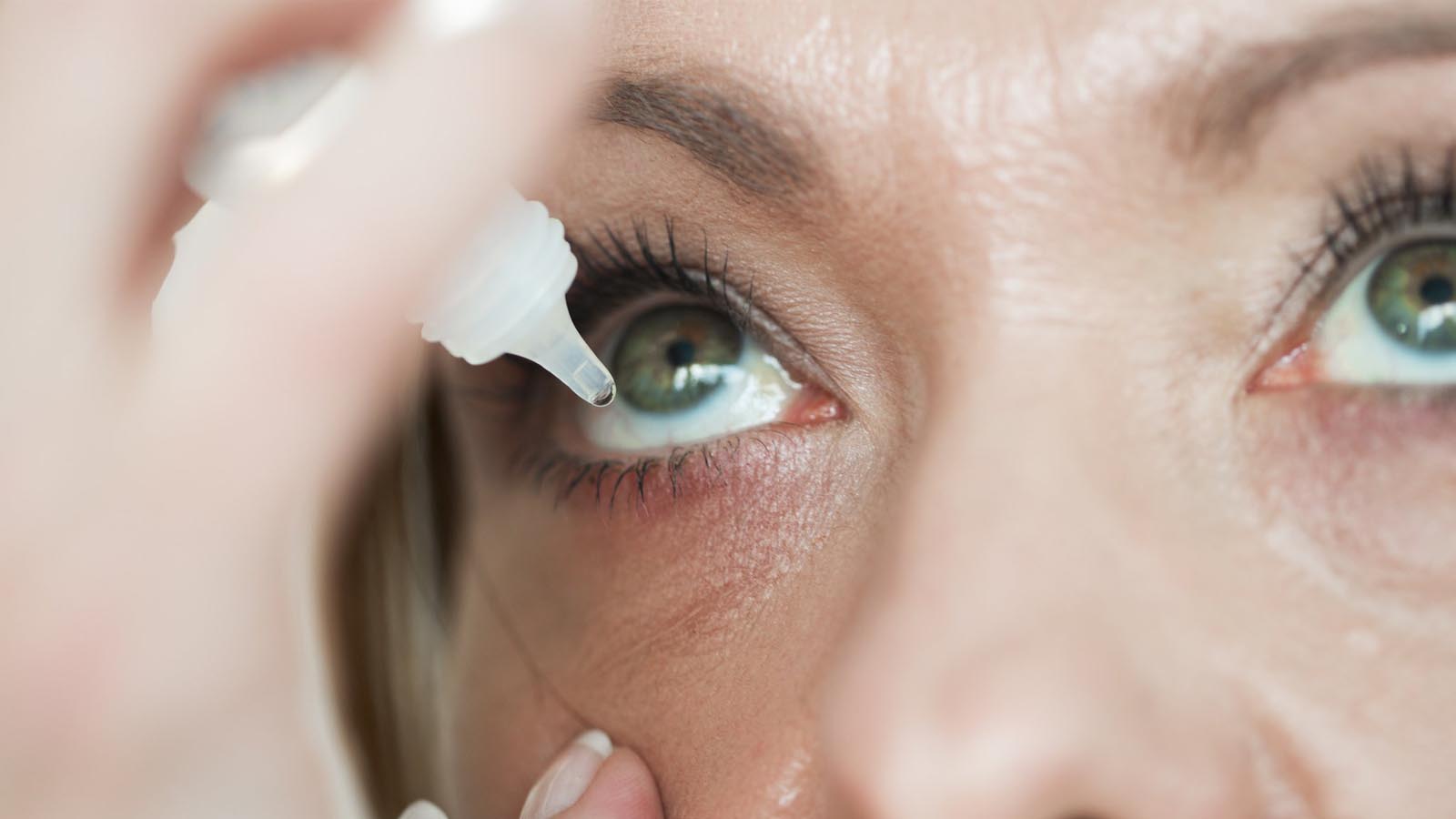
More than 10 different brands of artificial tears have been recalled. Most cases have been linked to EzriCare and Delsam Pharma eye drops, made by India-based Global Pharma Healthcare.
According to the CDC, the eye drops were contaminated with an antibiotic-resistant form of Pseudomonas aeruginosa, an aggressive bacterium.
Pseudomonas are a type of bacteria found in the environment, with P. aeruginosa being the most common to cause infections in humans.
The infection is common health care settings and spreads from improper hygiene either due to unclean hands or medical equipment and surfaces not being properly cleaned.
P. aeruginosa is resistant to multiple types of antibiotics and has caused about 32,600 infections among U.S. hospitalized patients and an estimated 2,700 deaths, according to the CDC.
The strain that has been linked to the outbreak, however, had never been reported in the United States before, the CDC stated in its update.
As of March 14, 68 people across 16 states have been infected with P. aeruginosa. Of those cases, 37 have been linked to four health care clusters.
“Testing of opened product identified the outbreak strain in bottles of EzriCare Artificial Tears that were obtained from two states,” the CDC told ABC News in a statement. “Testing of unopened product to evaluate for intrinsic contamination is ongoing by [the U.S. Food and Drug Administration].”
Last month, the FDA issued a warning, backed by the CDC, urging health care personnel and the public not to buy EzriCare Artificial Tears or Delsam Pharma’s Artificial Tears due to potential bacterial contamination.
After the warning, Global Pharma Healthcare issued a voluntary recall of both products, notifying distributors and advising wholesalers, retailers and customers who have the products to stop usage.
Not long after, the FDA also recommended that Global Pharma recall Delsam Pharma’s Artificial Eye Ointment, which the company agreed to. So far, no reports of infections have been linked to this product.
The CDC has warned anyone with symptoms of an eye infection who used EzriCare or Delsam Pharma eye drops to seek medical care immediately.
Such symptoms include yellow, green, or clear discharge from the eye; eye pain or discomfort; red eyes or eyelids; feeling of something in the eye; increased sensitivity to light; and blurry vision.
“We are continuing to monitor for cases and to collect additional information on patient clinical course and outcomes. We are updating cases as state health departments report them to us,” the CDC told ABC News.














Why do we need to sell eye drops, or any other medications for that matter, that were made in India? America is perfectly poised to manufacture and sell its own products. Purveyors need to get smarter about where they are sourcing products, and American manufacturers need to step up with any products countries like India, and better yet China, are manufacturing which places a dependency on our nation and puts control in the hands of nations like China.
they also need to put a stop to allowing any foreign entity to buying American land, china buying up all our farm land too
That too! (And the secret Chinese police organizations here and birthing tourism!)
Why are these products not produced in this country? Importation of any drug should be banned. At a time when country’s throughout the world want to destroy the USA we find out that drugs causing extreme health problems are being brought in from those places. That has to be the most underhanded attack on Americans to date.
It doesn’t surprise me anymore that we’d be using a third world country, India based pharmaceutical company. Seriously?
I thought we were only focused on made in China.